SENTI-202 for the Treatment of Acute Myeloid Leukemia (AML)
SENTI-202 for Acute Myeloid Leukemia
What is SENTI-202?
Cellular Backbone: Selected healthy adult donor derived NK cells
AML: A Significant Unmet Medical Need
Almost 10% of new cancer cases in the United States each year are hematologic malignancies, including leukemia, lymphoma and myeloma. AML is a type of acute leukemia characterized by an accumulation of malignant immature white blood cells. It is the most common type of acute leukemia in adults, constituting 80% to 85% of cases, and is the second most common—as well as the deadliest—in children. AML has a poor prognosis with a 5 year survival rate of 31.7% in the United States at diagnosis.
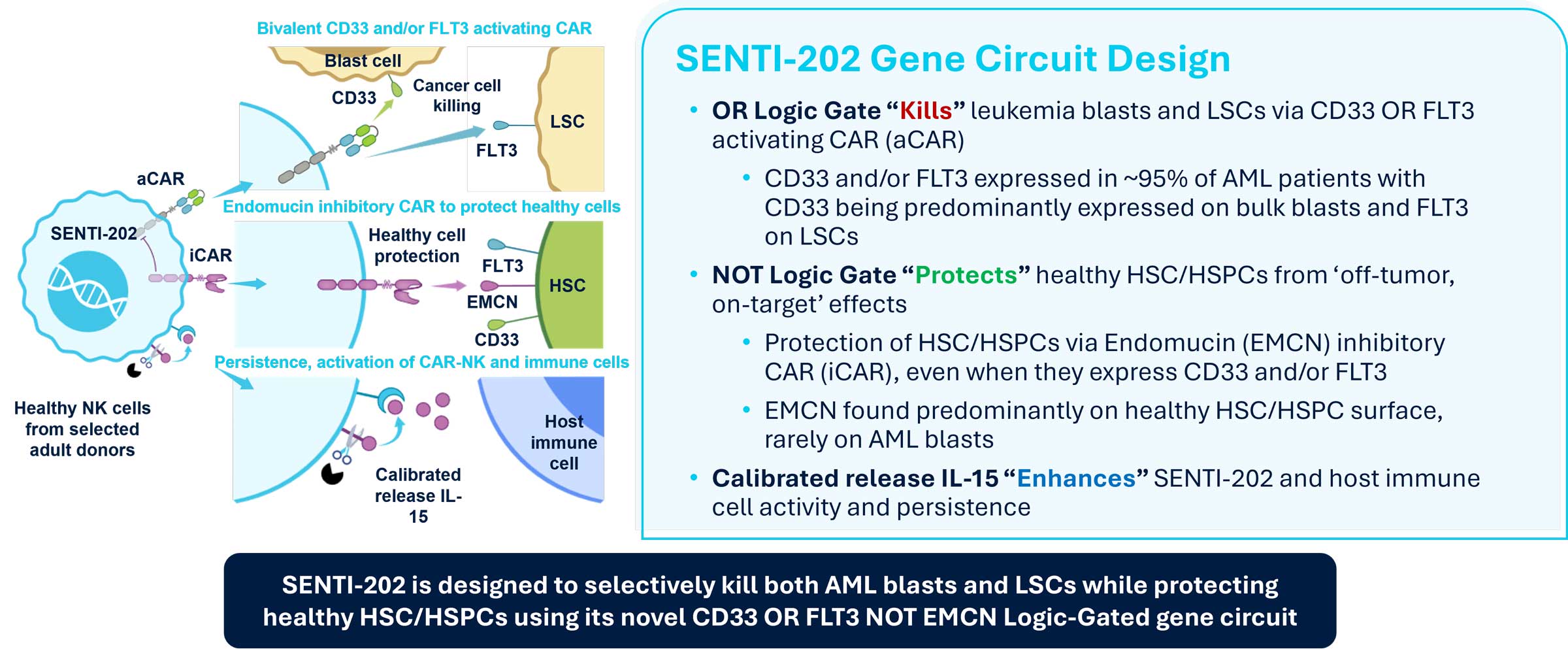
SENTI-202: First-in-Class Off-the-Shelf Logic-Gated Selective CD33 OR FLT3 NOT EMCN CAR NK Cell Therapy
SENTI-202 is our lead product candidate designed to selectively target and eliminate CD33 and/or FLT3-expressing hematologic malignancies, such as AML and myelodysplastic syndrome (“MDS”), while sparing healthy bone marrow cells. SENTI-202 has three main components. First, SENTI-202 contains an OR GATE (providing a “kill” signal), which is an activating CAR that recognizes CD33 and/or FLT3. By targeting either or both of these antigens, SENTI-202 is designed to effectively kill both leukemic blasts and leukemic stem cells, which constitute a difficult-to-eradicate reservoir of AML disease.
Second, SENTI-202 contains a NOT GATE (providing a “protect” signal), which is an inhibitory CAR that is designed to recognize healthy cells and protect those healthy cells from being killed, even if they were to express CD33 and/or FLT3, thus potentially widening the therapeutic window.
Third, SENTI-202 contains calibrated-release IL-15 (providing an “enhance” signal), which is designed to significantly increase cell persistence, expansion and activity of both the CAR-NK cells and host immune cells. The NK cells used to manufacture SENTI-202 are sourced from selected healthy adult donors.
Additionally, SENTI-202 has received Orphan Drug Designation from the US FDA for the treatment of relapsed/refractory hematologic malignancies including AML.
Intelligently Designed CAR-NK May Overcome Limitations of Current Therapies Against AML
SENTI-202 Phase 1 Clinical Trial in AML: Currently Enrolling
SENTI-202 Clinical Trial Design
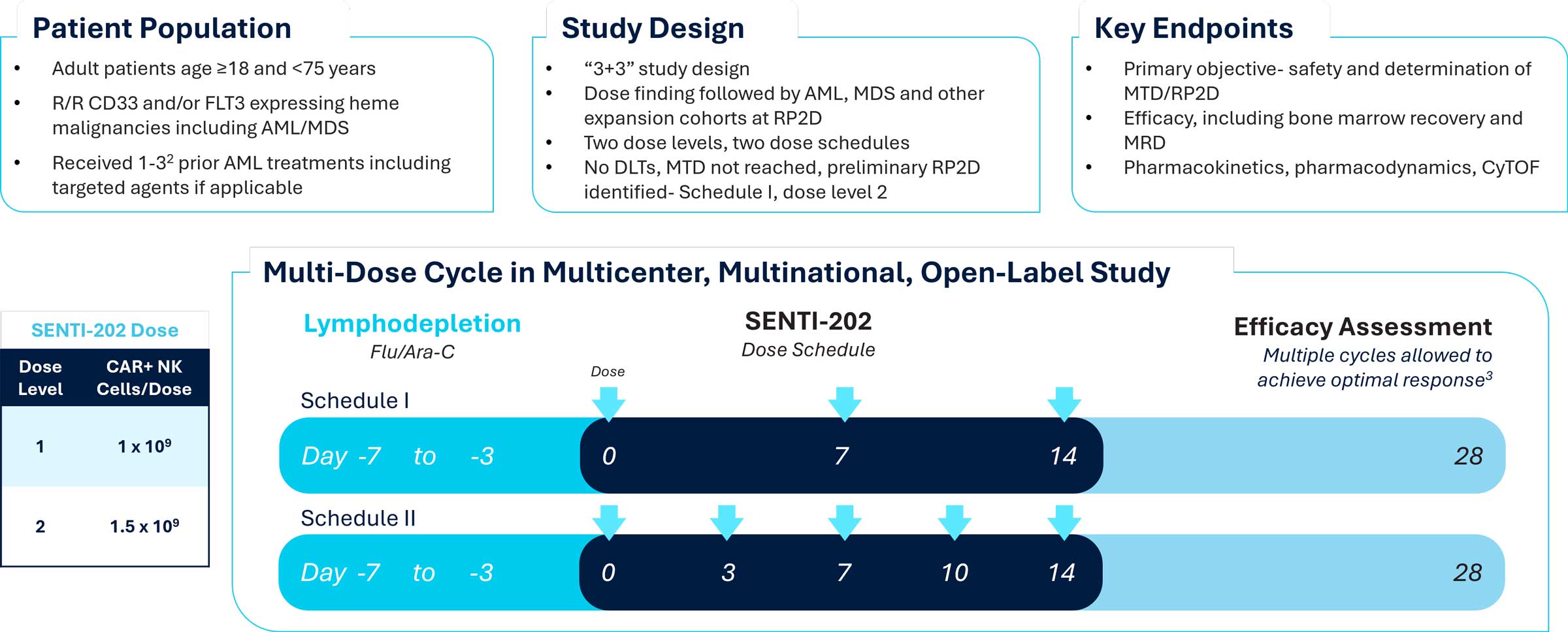
The Phase 1 clinical trial of SENTI-202 (NCT06325748) is enrolling across multiple sites in the United States and Australia. The study evaluates two dose levels of SENTI-202 (1 billion and 1.5 billion CAR NK cells per dose) administered in either a 3 dose regimen (Schedule 1: Days 0, 7, 14) or a 5 dose regimen (Schedule 2: Days 0, 3, 7, 10, 14) of a 28-day cycle following a fludarabine/cytarabine (Ara-C) based lymphodepletion in patients with relapsed or refractory CD33 and/or FLT3 hematologic malignancies including AML. Multiple cycles are permitted per protocol.
Demonstrates Positive Preliminary Clinical Results in the Treatment of Patients with Relapsed/Refractory AML
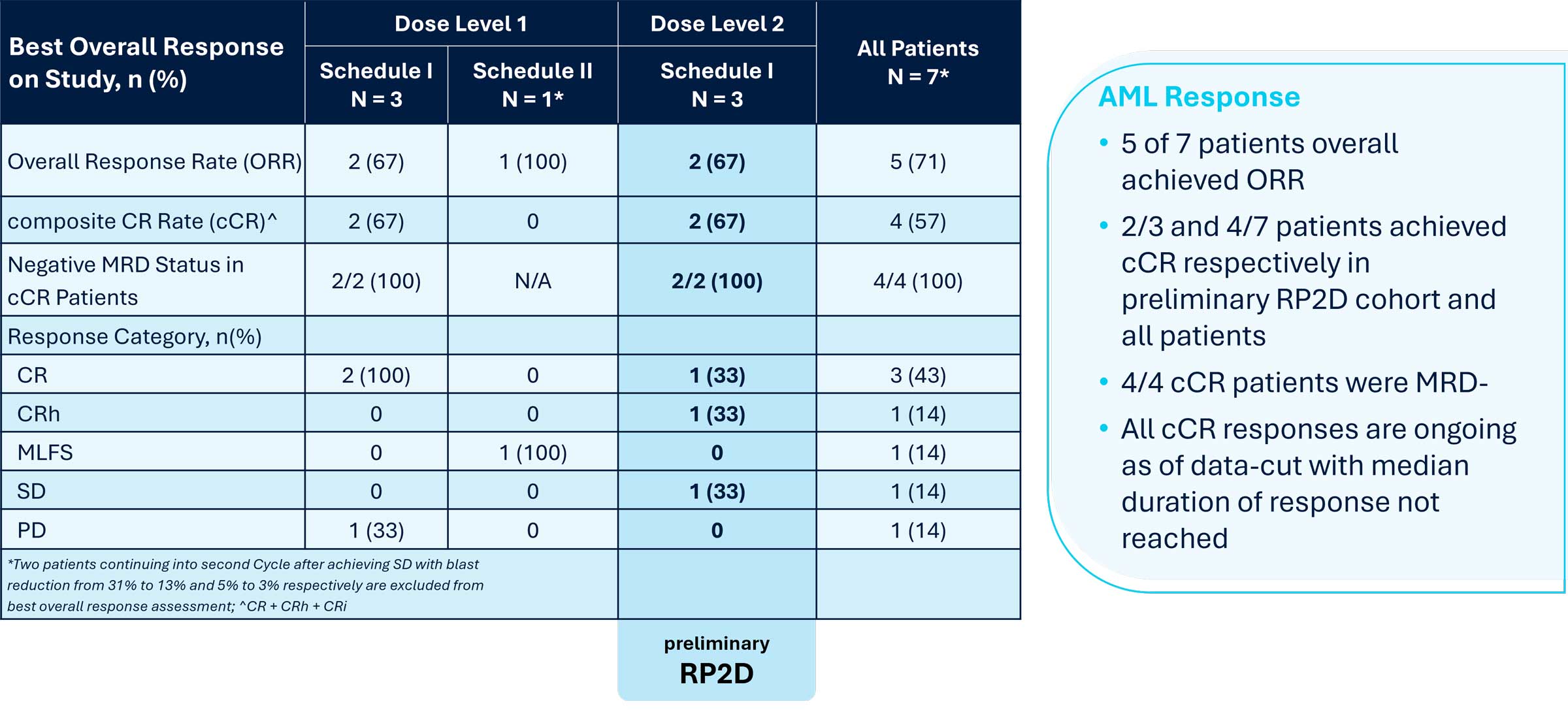
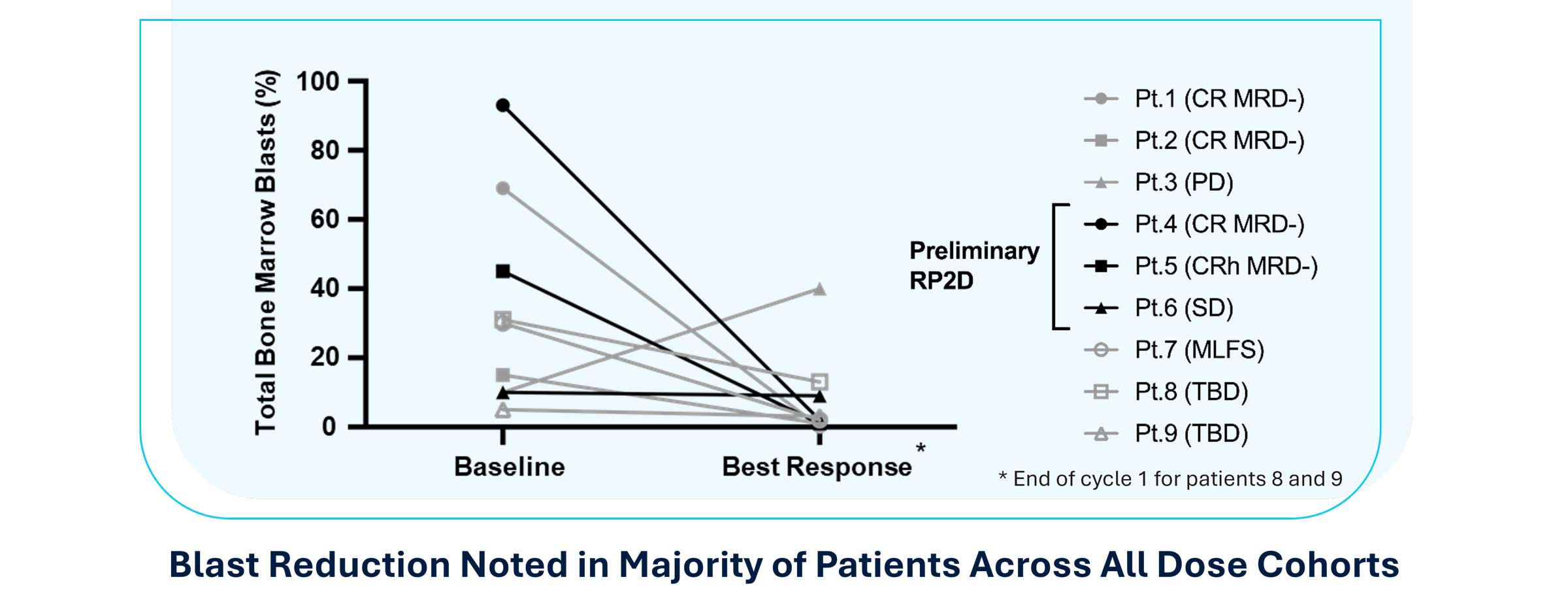
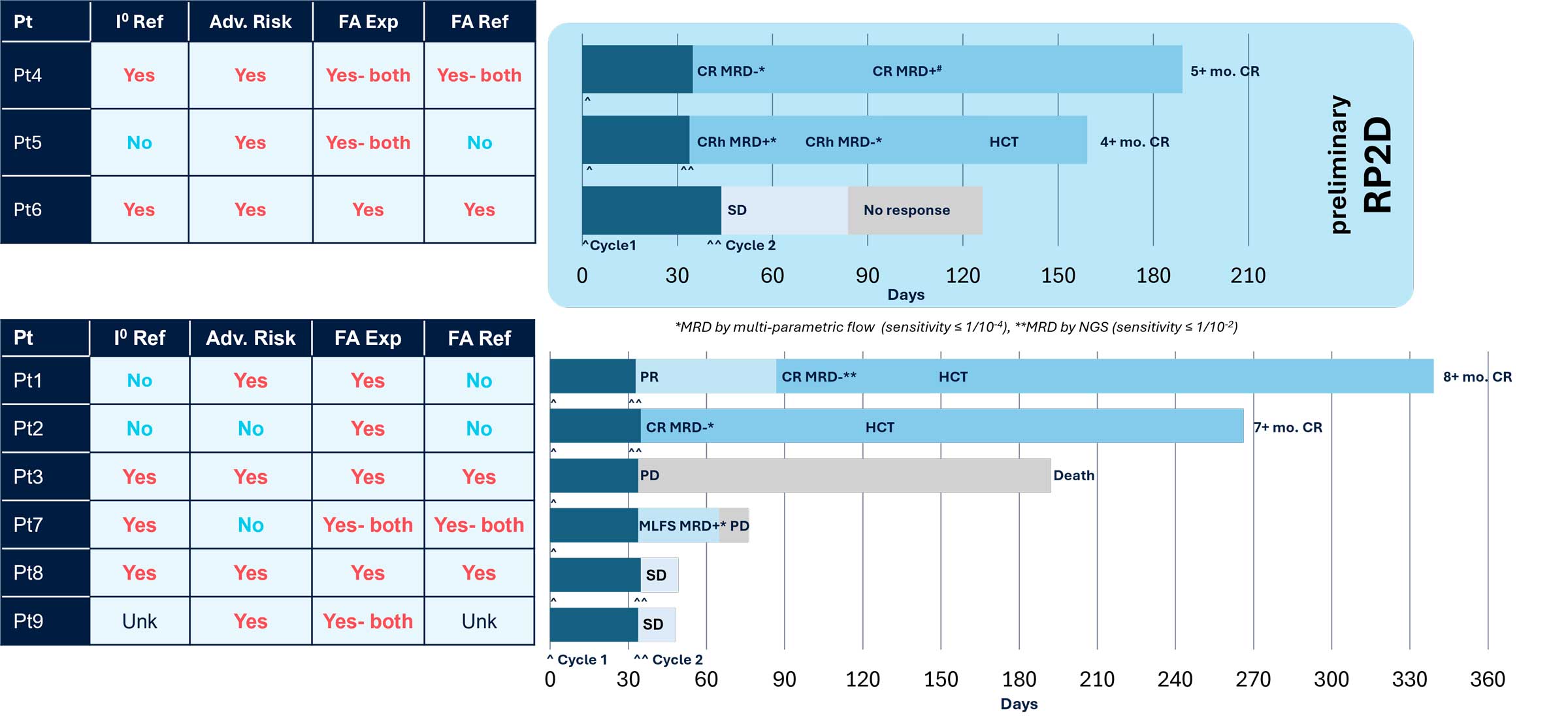
Positive preliminary efficacy results from the ongoing Phase 1 trial as well as correlative data from patients and preclinical data supporting Logic Gate mechanism of action were presented at the 2025 American Association for Cancer Research (AACR) Annual Meeting. The presented data demonstrated SENTI-202 is well-tolerated with no dose limiting toxicities and a maximum tolerated dose was not reached. The preliminary recommended Phase 2 dose (RP2D) was identified based on the totality of clinical data, including efficacy, as 1.5 x 109 CAR NK cells administered on Days 0,7,14 in 28-day Cycles, i.e. Schedule 1 following lymphodepleting chemotherapy. 2 of 3 patients in the preliminary RP2D cohort achieved a composite Complete Remission (cCR); 5 of the 7 best overall response evaluable patients achieved an ORR (cCR + morphologic leukemia-free state) outcome and 4 of the 7 achieved cCR (3 CR with full hematologic recovery, and 1 CRh (CR with partial hematologic recovery)). 4 of 4 cCR patients were MRD- (Measurable Residual Disease Negative) as assessed by local standard of care. All cCR patients were continuing in remission with follow-ups ranging from 4+ to 8+ months ongoing at the time of the AACR presentation.
Responses Observed Across All Dose Cohorts
Rapid Bone-Marrow Blast Reduction Observed Across All Dose Cohorts
Early Deep Responses Noted Across Dose Levels with Durability 8+ Months
Clinical Trial Access
We are committed to ensuring the safety of the patients who take part in our clinical trials and to upholding the highest ethical, scientific and clinical standards in all our research initiatives.
Clinical studies are an important part of the drug discovery process and a critical research tool for advancing patient care and bringing new medicines to patients and families who need them. Enrollment in one of our clinical studies is the primary way in which we can provide patients access to our investigational drugs prior to their potential approval by regulatory authorities, such as the United States Food and Drug Administration (FDA). Clinical studies are required to demonstrate that an investigational medicine meets necessary safety and effectiveness standards before it is approved and becomes commercially available to the public. Patients who participate in clinical studies help advance new scientific discoveries and make better future treatments possible.
Senti-sponsored clinical trials are designed and conducted in accordance with applicable laws and regulations, as well as recognized medical and ethical standards so that the health, well-being and safety of research participants are protected.
For more information about the SENTI-202 clinical trial, please click here.
If you are a patient or an investigator interested in one of our clinical trials, please e-mail info@sentibio.com. Thank you for your interest in Senti Bio!
Strategic Manufacturing Capabilities Enable Production of Off-The-Shelf Cell Therapies
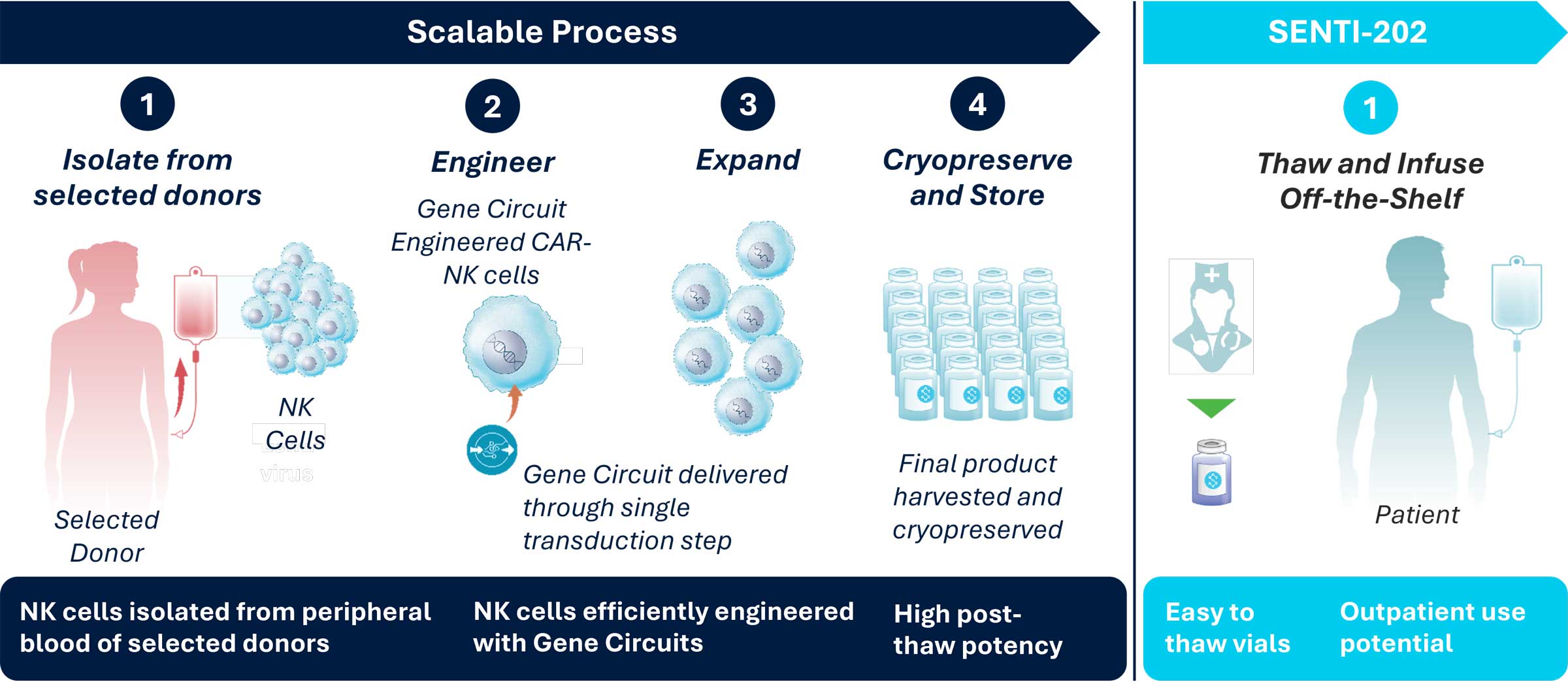
Our preferred cell source for our SENTI-202 product is peripheral blood NK cells from selected healthy adult donors because of their inherent anti-AML activity, this cell source allows us to immediately leverage an established supply chain, a mature GMP process and extensive clinical experience to develop our next generation CAR-NK cell therapies.
We have the ability to control the quality and supply of our off-the-shelf CAR-NK cell therapy product candidates for clinical studies and ultimately commercialization. We select healthy donors to maximize clinical activity, manufacturability and other features.
A key potential advantage of off-the-shelf cell therapies, versus autologous products that use each patient’s own cells, is the ability to manufacture large batches of drug product from healthy donor cells that can be produced in advance of clinical use stored in frozen vials, and shipped on-demand for patient use eliminating waiting times for patients. Upon commercialization, if our cell therapies are approved, we expect to be able to make these treatments broadly accessible in an off-the-shelf manner to cancer patients.